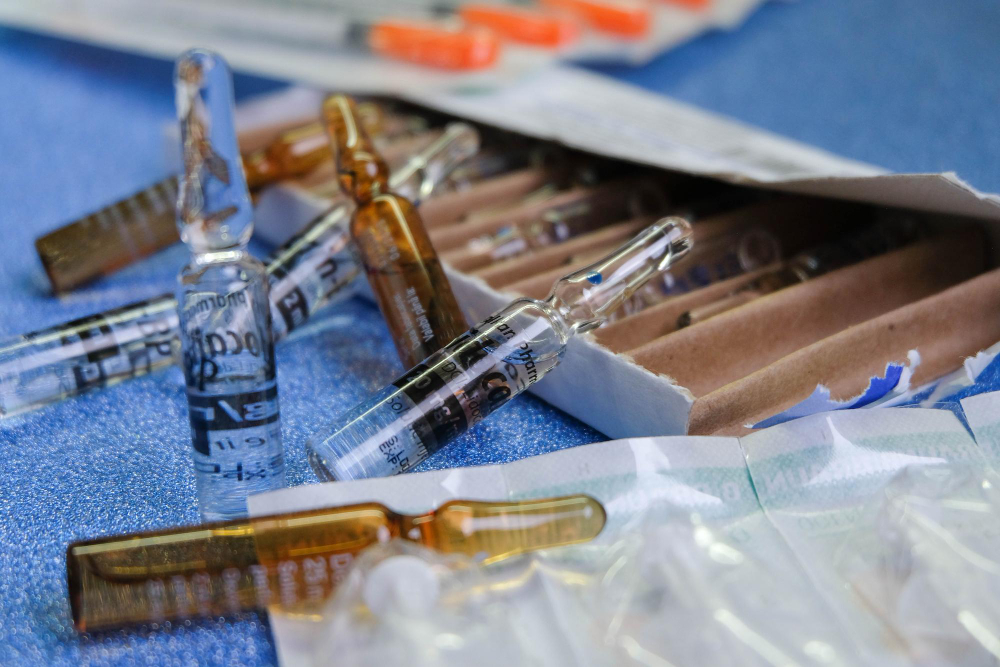
Safety in body modification relies heavily on protocols that often go unnoticed by the client. While the skill of the practitioner and the quality of the jewelry are visible factors, the integrity of the sterile packaging surrounding the piercing needle is the primary defense against infection. Sterile packaging is not merely a wrapper; it is a engineered barrier system designed to maintain sterility from the point of manufacturing until the moment of use. Understanding the standards governing these packages is essential for professional studios, health inspectors, and informed clients who prioritize safety above all else.
The consequences of compromised packaging extend beyond minor irritation. Breaches in sterile barriers can introduce bloodborne pathogens, bacteria, and environmental contaminants directly into open wounds. Regulatory bodies and industry organizations have established rigorous guidelines to mitigate these risks. These standards dictate everything from the materials used to construct the pouch to the chemical indicators printed on the surface. Compliance with these protocols ensures that the needle remains aseptic, protecting both the client and the practitioner from preventable health complications.
Regulatory Frameworks and Compliance Requirements
The production and distribution of piercing needles fall under strict medical device regulations. In the United States, the Food and Drug Administration (FDA) classifies piercing needles as medical devices, subjecting them to specific manufacturing and labeling controls. Manufacturers must adhere to Quality System Regulations, which require documented processes for design, production, and packaging. This oversight ensures that every needle leaving a facility meets consistent safety benchmarks. Non-compliance can result in recalls, legal liability, and severe health risks for end users.
Internationally, the International Organization for Standardization (ISO) provides the foundational technical standards for sterile medical device packaging. Specifically, ISO 11607 outlines the requirements for materials, sterile barrier systems, and packaging systems. This standard ensures that the packaging allows for sterilization while maintaining sterility until the point of use. Adherence to ISO 11607 is often a prerequisite for selling products in various global markets, signaling that the packaging has undergone validation testing for seal strength and microbial barrier properties.
Occupational safety is another critical component of the regulatory landscape. The Occupational Safety and Health Administration (OSHA) enforces the Bloodborne Pathogens Standard, which mandates how sharps and sterile items must be handled in the workplace. While this primarily protects workers, it indirectly safeguards clients by ensuring that sterile items are not contaminated during storage or handling. Studios must maintain an environment where sterile packaging is protected from moisture, dust, and physical damage to remain compliant with workplace safety laws.
Professional industry groups also contribute to these standards by setting best practices that often exceed minimum legal requirements. The Association of Professional Piercers (APP) publishes annual standards that member studios must follow to maintain membership. These guidelines frequently reference specific packaging integrity checks and sterilization verification methods. By aligning with such organizations, studios demonstrate a commitment to higher levels of safety and professionalism, providing clients with assurance regarding the hygiene protocols in place.
Materials and Design of Sterile Barrier Systems
The materials used in sterile packaging are selected for their ability to withstand sterilization processes while preventing microbial ingress. Common materials include medical-grade paper, Tyvek, and specialized plastic films. Each material offers distinct advantages regarding breathability, durability, and compatibility with different sterilization methods. For instance, paper-plastic pouches are widely used because the paper side allows sterilizing agents to penetrate, while the plastic side provides a transparent window for visual inspection of the needle inside.
Tyvek, a flash-spun high-density polyethylene fiber, is highly regarded in the medical industry for its strength and microbial barrier properties. It is resistant to tears and punctures, which is crucial when handling sharp objects like needles. When used in packaging, Tyvek ensures that the sterile barrier remains intact even under rough handling conditions. Manufacturers often choose this material for rigid containers or heavy-duty pouches where extra protection is necessary during shipping and storage.
The design of the seal is just as important as the material itself. Heat seals are the most common method for closing sterile pouches, creating a continuous bond that prevents bacteria from entering. The width of the seal channel is regulated to ensure sufficient strength. If a seal is too narrow, it may fail under stress; if it is too wide, it may interfere with the sterilization process. Quality control measures during manufacturing involve testing random samples to verify that the seal strength meets the minimum requirements set forth by AAMI (Association for the Advancement of Medical Instrumentation).
Transparency and labeling are integral design features. The packaging must allow the user to identify the needle gauge, length, and type without opening the sterile barrier. Additionally, external indicators must be visible to confirm that the package has undergone sterilization. Clear labeling reduces the risk of using the wrong instrument and ensures that expired or compromised packages are easily identified and discarded. This level of clarity is essential in high-volume studio environments where efficiency must not compromise safety.
Sterilization Methods and Packaging Compatibility
Sterile packaging must be compatible with the specific sterilization method used to process the needles. The three most common methods are Ethylene Oxide (EO) gas, Gamma Radiation, and Steam Autoclaving. Each method interacts differently with packaging materials, necessitating specific design considerations. Manufacturers must validate that their packaging maintains integrity and sterility after exposure to the chosen sterilization agent.
Ethylene Oxide sterilization is frequently used for pre-sterilized disposable needles. EO gas penetrates packaging materials effectively, making it suitable for porous materials like medical paper. However, the packaging must allow for gas penetration during the cycle and subsequent aeration to remove residual gas. Improper packaging can trap gas, leading to safety hazards, or prevent penetration, resulting in non-sterile products. Regulatory guidelines require strict monitoring of EO levels to ensure patient safety.
Gamma radiation is another method used for industrial sterilization of single-use needles. This process involves exposing the packaged needles to high-energy photons. Packaging materials used for gamma sterilization must be resistant to radiation degradation. Some plastics may become brittle or discolored when exposed to high doses of radiation, compromising the barrier. Manufacturers conduct testing to ensure that the material properties remain stable post-irradiation, maintaining the seal integrity required for long-term storage.
In professional studios, needles may sometimes be sterilized on-site using steam autoclaves. In this context, the packaging must be permeable to steam while preventing post-sterilization contamination. Paper-plastic pouches designed for autoclaving feature specific chemical indicators that change color when exposed to steam and heat. It is critical that practitioners use pouches rated for autoclave use rather than standard storage bags, as improper materials can melt or fail to allow steam penetration, rendering the sterilization cycle ineffective according to CDC infection control guidelines.
Integrity Testing and Chemical Indicators
Verifying the sterility of a package before use is a critical step in the safety protocol. Chemical indicators are printed on the exterior of sterile packaging to provide visual confirmation that the item has been through a sterilization process. These indicators change color when exposed to specific conditions, such as heat, steam, or gas. While they do not guarantee sterility, they serve as a immediate check to identify packages that may not have been processed correctly.
Internal chemical indicators are sometimes placed inside the packaging for higher levels of assurance. These are essential for complex instruments or dense loads where steam or gas penetration might be questionable. For single-use piercing needles, external indicators are standard, but the integrity of the package itself is the primary indicator of safety. Practitioners are trained to inspect every package for signs of compromise before breaking the seal.
Physical integrity testing involves checking for tears, holes, or seal failures. A package that appears wet, crushed, or open should never be used. Moisture can wick bacteria into the sterile field through the packaging material, a phenomenon known as strike-through. Similarly, pinholes caused by the needle tip pressing against the interior wall can create a direct path for contaminants. Rigorous visual inspection is the first line of defense against using compromised equipment.
Expiration dates are another critical component of integrity management. Sterile packaging is validated for a specific shelf life, during which the barrier properties are guaranteed. Over time, materials can degrade, seals can weaken, and indicators may fade. Using needles past their expiration date violates safety standards and increases the risk of infection. Studios must implement inventory rotation systems, such as first-in-first-out (FIFO), to ensure that older stock is used before newer shipments, aligning with ISO 13485 quality management standards.
Storage and Handling Best Practices
Proper storage conditions are vital for maintaining the integrity of sterile packaging until the moment of use. Environmental factors such as humidity, temperature, and light can degrade packaging materials over time. High humidity can weaken paper bonds and promote microbial growth on the exterior of the package. Therefore, sterile supplies should be stored in a climate-controlled environment away from sinks, windows, or areas prone to moisture fluctuations.
Physical protection during storage is equally important. Sterile packages should not be stacked too high, as the weight can crush the packages at the bottom, compromising the seals. Sharp objects should never be stored loosely in drawers where they can puncture neighboring packages. Many professional studios utilize dedicated cabinets or bins designed to hold sterile pouches upright or in single layers to prevent physical damage. This organizational strategy minimizes the risk of accidental breaches before the package is even opened.
Handling protocols dictate that sterile packages should only be touched with clean, dry hands or gloved hands immediately before use. Dropping a sterile package on the floor renders it non-sterile, regardless of the integrity of the seal. The floor is considered a contaminated zone, and impact can create micro-tears invisible to the naked eye. If a package is dropped or suspected of contamination, it must be discarded and replaced with a new sterile unit. This zero-tolerance policy is a cornerstone of WHO injection safety practices.
Transportation of sterile items within the studio also requires care. Moving packages from storage to the procedure room should be done in a manner that protects them from environmental exposure. Open trays or containers used for transport should be clean and dedicated solely to sterile items. Mixing sterile and non-sterile items in the same container increases the risk of cross-contamination. Clear labeling of transport containers helps maintain the distinction between clean and dirty zones within the workspace.
Comparison of Packaging Types and Sterilization Methods
Different packaging configurations offer varying levels of protection and suitability for specific sterilization methods. Understanding the differences helps professionals select the right supplies for their operational needs. The following table outlines the key characteristics of common packaging types used for piercing needles.
| Packaging Type | Primary Material | Compatible Sterilization Method | Shelf Life Expectancy | Best Use Case |
|---|---|---|---|---|
| Paper-Plastic Pouch | Medical Paper / Poly Film | Steam Autoclave, EO Gas | 1–5 Years | On-site sterilization, single-use needles |
| Tyvek Pouch | Synthetic Fiber / Poly Film | EO Gas, Gamma Radiation | 3–5 Years | Pre-sterilized disposables, heavy-duty protection |
| Rigid Container | Medical Grade Plastic/Metal | Steam Autoclave, EO Gas | Indefinite (if seals intact) | Reusable instrument sets, high-volume studios |
| Blister Pack | Formed Plastic / Foil | Gamma Radiation, EO Gas | 2–5 Years | Retail single-use needles, individual sale |
| Wrap Material | Medical Grade Cloth/Paper | Steam Autoclave | 30 Days (event-related) | Custom instrument trays, specialized setups |
The data indicates that pre-sterilized disposable needles often utilize Tyvek or Blister Packs due to their compatibility with industrial gamma radiation and long shelf life. In contrast, studios that sterilize their own instruments rely heavily on Paper-Plastic Pouches compatible with steam autoclaves. Rigid containers offer durability but require careful maintenance of filters and seals to remain effective. Selecting the appropriate packaging depends on the sterilization equipment available and the volume of procedures performed.
Common Risks and Mitigation Strategies
Despite stringent standards, risks associated with sterile packaging can arise during the supply chain or within the studio. One common issue is the purchase of non-compliant products from unverified suppliers. Needles sourced from manufacturers that do not adhere to FDA or ISO standards may arrive in packaging that fails to maintain sterility. Mitigating this risk requires purchasing only from reputable distributors who can provide documentation of compliance and sterilization validation.
Another risk involves the misinterpretation of chemical indicators. A color change confirms exposure to sterilization conditions but does not guarantee the absence of all microorganisms. Relying solely on the indicator without considering the physical integrity of the package can lead to false security. Comprehensive safety protocols require both indicator verification and physical inspection. Regular training for staff on how to interpret these indicators correctly is essential for maintaining high safety standards.
Improper disposal of packaging also presents a risk. Once a needle is used, the packaging becomes contaminated with blood or bodily fluids. It must be disposed of according to medical waste regulations. Mixing sterile packaging waste with sharps waste or general trash can violate EPA medical waste guidelines. Studios should have clearly labeled bins for biohazard waste and ensure that all staff members are trained on proper segregation and disposal procedures to prevent environmental contamination.
Frequently Asked Questions
How can I tell if a piercing needle package is truly sterile?
Look for intact seals, no signs of moisture or tearing, and a chemical indicator that has changed to the correct color. The package should also be within its expiration date. If any of these factors are compromised, the sterility cannot be guaranteed.
Is it safe to use needles past their expiration date if the package looks fine?
No. The expiration date is based on validated testing that ensures the barrier material maintains its integrity over time. Using expired needles violates safety standards and increases the risk of infection, even if the package appears intact.
Can I sterilize needles at home using boiling water?
No. Boiling water does not achieve the temperatures required for sterilization and cannot penetrate sealed packaging effectively. Home sterilization methods are insufficient for breaking the skin and pose significant health risks. Professional autoclaves are required for true sterilization.
What should I do if I drop a sterile needle package on the floor?
The package should be discarded immediately. The floor is a contaminated surface, and the impact may have created micro-tears in the packaging that are not visible. Always open a new, verified sterile package for the procedure.
Do all piercing needles come pre-sterilized?
Most single-use needles purchased from professional suppliers come pre-sterilized in sealed packaging. However, some reusable instruments require on-site sterilization. Always check the labeling to determine the sterilization status and requirements.
How long does sterility last once the package is opened?
Sterility is compromised the moment the package is opened. The needle must be used immediately or discarded. There is no safe way to re-store an opened needle for later use.
Are there different packaging standards for different body piercings?
The packaging standards remain consistent regardless of the piercing location. However, the needle gauge and length will vary. The integrity requirements for the packaging do not change based on the procedure type.
What is the difference between sanitized and sterile?
Sanitized means the number of bacteria has been reduced to a safe level, while sterile means the complete absence of all microbial life. Piercing needles must be sterile, not just sanitized, to prevent infection in open wounds.
Can I reuse sterile packaging pouches for autoclaving?
No. Single-use pouches are designed for one sterilization cycle. Reusing them can compromise the material integrity and the chemical indicators, leading to potential sterilization failure.
Where should I store sterile needles in my studio?
Store them in a clean, dry, enclosed cabinet away from sinks and high-traffic areas. Avoid storing them on the floor or in areas where they could be crushed or exposed to moisture.
Conclusion and Future Outlook
The standards governing sterile packaging for piercing needles represent a critical intersection of material science, regulatory compliance, and public health. These protocols ensure that every needle used in a professional setting provides a safe pathway for body modification without introducing unnecessary health risks. From the manufacturing floor to the studio cabinet, every step in the lifecycle of the packaging is designed to preserve the aseptic condition of the instrument. Adherence to these standards is not optional; it is a fundamental requirement for ethical practice.
As technology advances, packaging materials continue to evolve. Innovations in smart packaging may soon offer digital indicators that provide real-time data on sterility status. Enhanced materials could offer greater resistance to environmental factors while remaining eco-friendly. However, the core principles of integrity, validation, and proper handling will remain unchanged. Professionals must stay informed about these developments to maintain the highest levels of safety.
For studio owners and practitioners, investing in high-quality packaging and adhering to strict storage protocols is an investment in client trust and legal protection. Regular audits of supply chains, storage conditions, and handling procedures ensure that safety standards are consistently met. Clients benefit from this diligence through reduced healing times and lower risks of complications. Ultimately, the invisible shield of sterile packaging is what allows the art of piercing to be practiced safely and sustainably.
Prioritizing these standards fosters a culture of safety within the industry. It encourages transparency and accountability, ensuring that body modification remains a respected and safe practice. By understanding and respecting the science behind sterile packaging, professionals uphold their duty of care. The future of the industry depends on this commitment to excellence, ensuring that every procedure is performed with the utmost regard for human health and safety.
